Introduction
A failed nucleic acid extraction often starts before the protocol — it starts at preservation. Whether a tissue sample arrives fresh frozen or formalin-fixed paraffin-embedded (FFPE) directly shapes what you can extract, how much, and how well it performs in downstream assays like NGS, PCR, or RNA sequencing. Poor preservation choices translate to failed extractions, degraded libraries, and inconclusive mutation calls that can derail studies or clinical decisions before they begin.
Fresh frozen tissue remains the gold standard for nucleic acid integrity — yet the vast majority of archived clinical specimens are FFPE. Peer-reviewed estimates put globally NGS-suitable FFPE solid tumor blocks at 50 to 80 million, representing an enormous resource for retrospective research that labs cannot ignore.
Understanding when each method delivers reliable results — and when they are genuinely interchangeable — helps laboratories navigate real trade-offs between sample accessibility, infrastructure costs, and molecular data quality.
TL;DR
- Fresh frozen tissue preserves DNA and RNA in native, intact form, making it the preferred input for WGS, WES, and RNA-Seq
- FFPE tissue offers room-temperature storage and access to decades-old archives, but formalin fixation causes DNA fragmentation, cross-linking, and RNA degradation
- Studies show >94% variant-level concordance between FFPE and fresh frozen results in targeted mutation panels when optimized protocols are used
- FFPE-optimized extraction workflows make archival tissue viable when fresh samples are unavailable
Fresh Frozen Tissue vs FFPE: At a Glance
| Feature | Fresh Frozen Tissue | FFPE Tissue |
|---|---|---|
| Preservation Method | Snap-frozen in liquid nitrogen | Formalin-fixed, paraffin-embedded |
| Storage Requirements | −80°C or below (liquid nitrogen vapor) | Room temperature |
| DNA Quality | High-molecular-weight (>50,000 bp); intact genomic DNA | Fragmented (127-177 bp median); chemically modified |
| RNA Quality | High integrity (RIN 7-10) | Degraded (low RIN, but DV200 ≥30% usable) |
| Typical Applications | WGS, WES, RNA-Seq, proteomics | Histopathology, IHC, targeted NGS panels |
| Global Availability | Limited (requires specialized biobanks) | Extensive (50-80 million+ archived blocks) |
| Storage Cost | High (power, liquid nitrogen, maintenance) | Low (room temperature, stable for decades) |

Real-world performance depends heavily on sample handling at collection, fixation duration (for FFPE), and extraction method. In a 2020 paired colorectal cancer study, fresh frozen tissue consistently yielded higher total sequencing coverage than FFPE — median 3,739 reads vs. 2,814 reads — yet both achieved high clinical concordance for mutation detection.
Method selection ultimately comes down to your application: fresh frozen for discovery-phase genomics requiring intact nucleic acids, FFPE when retrospective archives or histological context are the priority.
What is Fresh Frozen Tissue?
Fresh frozen tissue refers to biological specimens — typically biopsies or surgically resected tissue — that are rapidly cooled by immersion in liquid nitrogen through a process called "snap-freezing" or "flash-freezing." The samples are then stored at ≤ −80°C to halt enzymatic activity and preserve molecular integrity. Cold ischemia time (time from excision to freezing) should be minimized to less than 20 minutes, but no more than 1 hour, as delays can compromise RNA and protein quality.
When executed correctly, flash-freezing preserves several molecular classes in near-native form:
- Intact high-molecular-weight DNA suitable for WGS, WES, and long-read sequencing (>50,000 bp fragments)
- High-quality RNA with RIN scores typically 7-10, enabling RNA-Seq, gene expression profiling, and transcriptomic studies
- Native, non-denatured proteins useful for proteomic and biochemical analyses
Practical challenges:
- Requires liquid nitrogen availability near the operating room
- Dedicated −80°C ultralow-temperature freezers with continuous power supply
- Vulnerability to power outages and freeze-thaw cycles (each cycle leads to macromolecular degradation and reduced RNA integrity)
- Rigorous chain-of-custody documentation
- High costs for large-scale biobanking
These hurdles explain why fresh frozen biobanks hold far fewer samples than FFPE collections. Fresh frozen tissue can also be embedded in OCT (Optimal Cutting Temperature) compound for cryosectioning, enabling immediate histological analysis during surgery — though OCT significantly inhibits PCR amplification, particularly for fragments >300 bp, limiting downstream molecular applications.
Despite these constraints, fresh frozen tissue remains the gold standard for several high-demand molecular workflows.
Use Cases of Fresh Frozen Tissue
- Whole genome sequencing (WGS) and whole exome sequencing (WES) requiring high-molecular-weight DNA
- RNA-Seq and microarray-based transcriptomics where RNA integrity is critical
- Proteomics studies requiring native protein conformation
- Long-read sequencing workflows (Oxford Nanopore, PacBio)
- Intraoperative frozen sections for real-time surgical decisions, such as margin assessment during tumor resection
- Precision oncology biobanks where retrospective access to high-quality nucleic acids is essential
What is FFPE Tissue?
FFPE (formalin-fixed paraffin-embedded) tissue is the standard preservation method used in pathology for decades. Tissue is immersed in 10% neutral buffered formalin for 18–72 hours, which cross-links proteins and halts cellular processes — chemically "fixing" the sample. The fixed tissue is then dehydrated in ethanol, cleared in xylene, and embedded in molten paraffin wax that solidifies into a block.
These blocks store at room temperature for decades, making FFPE the primary sample type held in hospital pathology archives and biobanks worldwide.
The key trade-off
Formalin fixation preserves cellular architecture and morphology exceptionally well — making FFPE the gold standard for histopathology, H&E staining, and immunohistochemistry. However, the same chemical cross-linking process degrades nucleic acids:
- DNA becomes fragmented into smaller pieces (median insert sizes 127–177 bp)
- Chemical modification through deamination causes artifactual C>T and G>A transitions
- RNA is substantially degraded, complicating transcriptomic analysis
FFPE Quality Is Not Uniform
Fixation time, tissue thickness, temperature during fixation, formalin concentration, and paraffin storage duration all affect recoverable nucleic acid quality. Fixation for 48 hours or more significantly increases deamination events and decreases mapped read percentages. Specimens stored for many years yield lower-quality DNA and RNA, and the lack of standardised fixation protocols across institutions compounds this variability.
Despite these limitations, the scale of available material is difficult to overstate. Research estimates suggest 50 to 80 million FFPE solid tumour specimens are potentially suitable for NGS analysis globally, many with linked long-term clinical data covering diagnosis, treatment regimen, and outcomes. This makes FFPE the most practical source for large-scale retrospective oncology research and real-world evidence studies.
Use Cases of FFPE Tissue
Primary applications:
- Histopathology and morphological assessment (H&E staining, IHC, immunofluorescence) for cancer diagnosis, grading, and staging
- Molecular testing including targeted mutation panels (KRAS, EGFR, BRAF, TP53) and NGS-based multi-gene panels for companion diagnostics
- DNA methylation analysis
- Retrospective biomarker studies leveraging decades-old archived specimens
FFPE is what pathology labs routinely produce, so most clinical NGS workflows are built around it — including companion diagnostics for targeted therapy selection. Laboratories have developed FFPE-optimised extraction and library preparation protocols specifically to work within its quality constraints.
DNA & RNA Extraction Quality: Head-to-Head
DNA Yield and Integrity
Fresh frozen tissue yields high-molecular-weight, intact DNA with significantly higher sequencing coverage depth. Research comparing paired fresh frozen and FFPE colorectal cancer samples found median total coverage of 3,739 reads (IQR: 2,148-5,866) for fresh frozen versus 2,814 reads (IQR: 1,784-3,936) for FFPE.
FFPE DNA is severely fragmented — median insert sizes frequently range from 127 bp to 177 bp, compared to >50,000 bp for fresh frozen. This fragmentation limits applications requiring long reads or high-fidelity amplification. However, short-amplicon PCR-based panels (<200 bp products) can achieve high concordance with fresh frozen results.
RNA Quality: The Critical Gap
RNA degradation in FFPE is far more severe than DNA degradation. RNA is susceptible to both enzymatic degradation during the delay before fixation and chemical modification during formalin fixation itself.
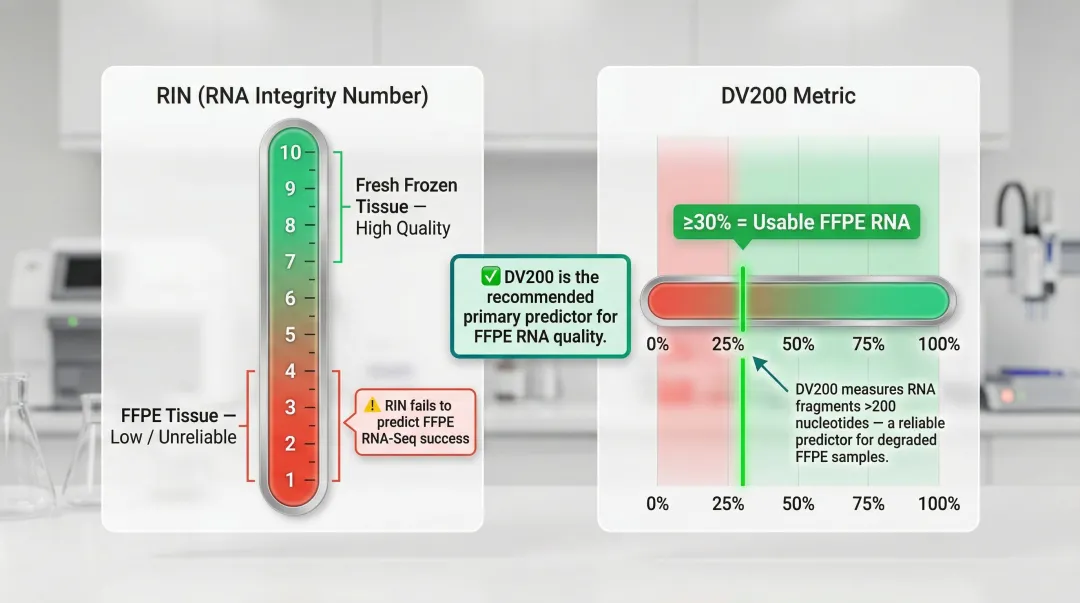
DV200: The FFPE RNA Quality Standard
RIN (RNA Integrity Number) scores fail to predict FFPE RNA-Seq success. Instead, the DV200 metric — the percentage of RNA fragments >200 nucleotides — strongly correlates with library yield and gene detection.
- Fresh frozen RNA typically achieves RIN scores of 7-10
- FFPE RNA often has low RIN but can still be usable when DV200 ≥30%
- Enhanced quality metrics for FFPE RNA recommend DV200 as the primary predictor
Mutation Concordance Data
A study comparing FFPE and fresh frozen tissue using a 22-gene NGS panel in 118 colorectal cancer patients found >94% concordance across all variants at the variant level, with gene-level concordance ranging from approximately 73.8% to 100%.
Critical caveat: FFPE-specific artifacts exist. Formalin induces C>T and G>A transitions (up to a 7-fold increase vs. frozen), particularly at CpG sites, confounding low-allele-frequency variant calling. These artifacts must be filtered bioinformatically.
FFPE is a viable alternative for targeted mutation detection, but it is not a direct replacement for fresh frozen when maximum sensitivity and specificity are required.
Extraction Method Matters
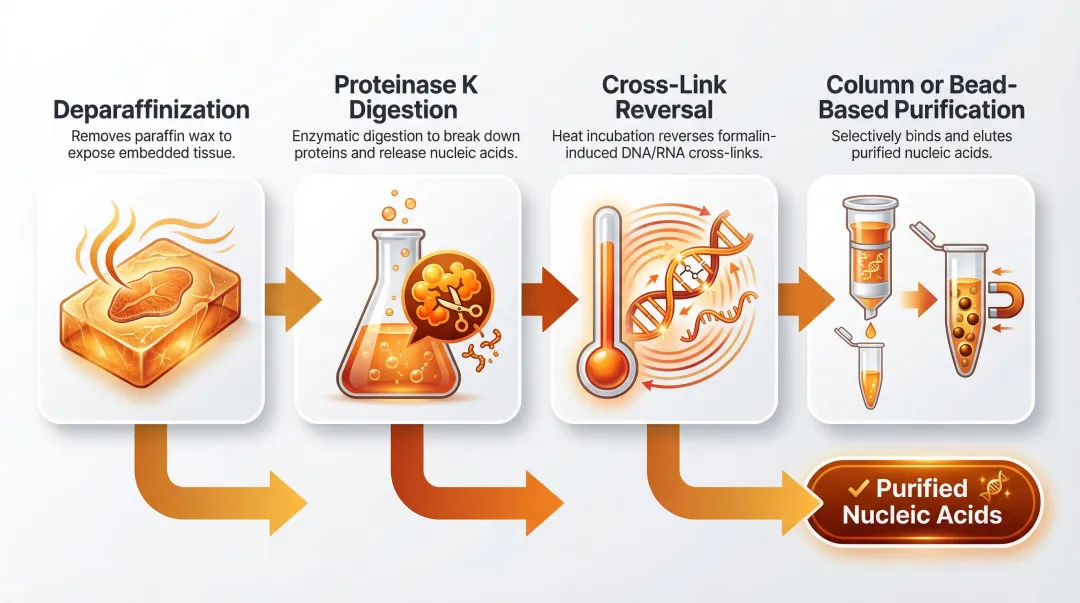
These concordance figures, however, assume an optimised extraction workflow. The quality of nucleic acids recovered from FFPE is dependent on each step in the process:
- Deparaffinization efficiency
- Proteinase K digestion time
- Cross-link reversal (heat incubation)
- Column or bead-based purification systems
Automated magnetic bead-based extraction platforms (such as Cambrian Bioworks' Manta system) can standardise the FFPE extraction process, reduce variability from manual handling, and improve reproducibility across samples. Manta completes DNA extraction in 28 minutes after lysis and processes 1–32 samples per run, with chemistry developed to address the crosslinking and fragmentation characteristic of FFPE material.
Downstream Application Suitability
Application Framework:
| Application | Fresh Frozen | FFPE |
|---|---|---|
| WGS/Long-Read Sequencing | Strongly favoured | Not recommended |
| Targeted NGS Panels (<200 bp) | Optimal | Viable with optimized protocols |
| RNA-Seq/Whole Transcriptome | Strongly preferred | Possible with FFPE-specific kits & DV200 ≥30% |
| IHC/Histopathology | Inferior morphology | Superior (gold standard) |
| Real-Time PCR (Hotspot Mutations) | Feasible | Feasible with appropriate kits |
Which Tissue Type Should Your Lab Choose?
Choose Fresh Frozen When:
- Your application demands maximum nucleic acid integrity
- You're conducting WGS, long-read sequencing, or RNA-Seq-based transcriptomics
- Detecting low-frequency variants at high sensitivity is critical
- You're running prospective research studies where sample collection can be planned
- Infrastructure (liquid nitrogen, −80°C freezers) is available
Choose FFPE When:
- Fresh tissue is unavailable (the reality for most retrospective clinical studies)
- You're validating biomarkers using archived specimens
- Companion diagnostic testing in routine pathology workflows is the goal
- The primary objective is morphological assessment (IHC, H&E)
- Working with millions of clinically annotated archived samples in hospital biobanks
Situational Nuance
When both options are theoretically available, consider that concordance studies demonstrate FFPE can reliably substitute for fresh frozen in targeted DNA mutation panels. However:
- Standardize fixation times (18-24 hours using 10% NBF)
- Validate extraction protocols for your specific FFPE sample age and tissue type
- Use bioinformatic filters to remove FFPE-specific artifacts
- For RNA applications, the gap between fresh frozen and FFPE remains significant — FFPE-specific RNA extraction and library prep solutions are essential to obtain usable data
Navigating fixation variability and protocol differences across sample types is where extraction chemistry matters most. Cambrian Bioworks' magnetic bead-based kits are validated for both FFPE and fresh frozen tissue, giving labs a single extraction platform that handles the protocol shifts these two sample types demand — without rebuilding workflows from scratch.
Frequently Asked Questions
What is the difference between fresh frozen tissue and FFPE tissue?
Fresh frozen tissue is snap-frozen in liquid nitrogen and stored at −80°C to preserve nucleic acid integrity, while FFPE tissue is chemically fixed in formalin and embedded in paraffin wax for room-temperature storage. The key practical difference is that fresh frozen yields higher-quality DNA and RNA while FFPE offers greater accessibility and long-term morphological preservation.
What does formalin-fixed paraffin-embedded (FFPE) mean?
FFPE is a two-step preservation process. Formalin fixation cross-links proteins to halt biological activity and preserve tissue structure; paraffin wax embedding then protects the specimen from degradation and enables thin sectioning for microscopy. FFPE blocks can be stored at room temperature for decades.
What is a fresh frozen sample?
A fresh frozen sample is biological tissue, typically a biopsy or surgical specimen, rapidly cooled by immersion in liquid nitrogen immediately after collection and stored at −80°C or below. This preserves DNA, RNA, and proteins in their intact, native state — making it the go-to choice for downstream molecular analysis such as sequencing and expression profiling.
What are the advantages and disadvantages of cryosections versus paraffin-embedded sections?
Cryosections offer rapid turnaround and preserve native protein antigenicity for IHC, but morphological detail is inferior to paraffin. Paraffin-embedded sections are the histopathology standard for morphology, though fixation denatures proteins and degrades nucleic acid quality.
Is a frozen section biopsy accurate?
Frozen section biopsies are accurate enough for intraoperative decision-making, achieving pooled sensitivity of 0.81 and specificity of 0.97 for breast-conserving surgery margins. However, for definitive pathological diagnosis or detailed molecular analysis, results are typically confirmed with permanent FFPE sections.
Can you paraffin embed frozen tissue?
It is possible, but not standard practice. Freezing prior to fixation introduces ice crystal artifacts that disrupt tissue architecture, resulting in inferior morphological quality compared to tissue processed fresh for FFPE embedding.


