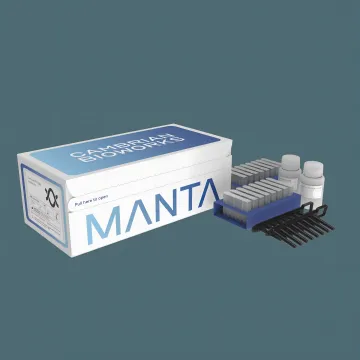
Cambrian FFPE gDNA Isolation Kit
- Fast 28-min workflow post-lysis
- 1 to 32 samples per run, flexible
- High-MW gDNA for NGS, PCR & more
ISO 13485 Certified
CE-IVD Mark
CDSCO IVD Approved
Cambrian Bioworks offers CDSCO-approved nucleic acid extraction kits designed for clinical diagnostics and research in India. The portfolio includes FFPE gDNA, whole blood gDNA, and blood RNA isolation kits, all powered by magnetic bead technology on the automated Manta platform for fast, reproducible, and high-quality results.





You can verify CDSCO approval by searching the Central Drugs Standard Control Organisation's official online database at cdsco.gov.in. For IVD products like diagnostic kits, look for a valid CDSCO IVD manufacturing license number. Cambrian Bioworks holds CDSCO IVD certification MFG/IVD/2025/000069, which is publicly verifiable through this portal.
Talk to our experts for custom solutions and tailored guidance.

FFPE gDNA and blood RNA kits support tumor mutation analysis, NGS-based cancer diagnostics, and blood cancer monitoring.

CDSCO-approved kits enable reliable gDNA and RNA extraction from whole blood for molecular diagnostics and clinical reporting.

High-molecular-weight DNA and high-RIN RNA outputs enable NGS, microarray, and transcriptomics workflows in research settings.
Whole blood gDNA and RNA kits support infectious disease testing, reproductive genetics, and genetic counseling applications.
Talk to our experts for custom solutions and tailored guidance.
All kits carry CDSCO IVD and CE-IVD certification, ensuring full regulatory compliance for clinical diagnostics in India and Europe.
Complete nucleic acid extraction in under 30–50 minutes with 56% lower cost per test and 74% faster turnaround than conventional methods.
From Tata Memorial Hospital to leading research institutes, Cambrian Bioworks is the trusted extraction partner across oncology, genetics, and diagnostics labs.
Submit your details and our team will respond within one business day with pricing, demo scheduling, and technical guidance.
For immediate assistance, feel free to give us a direct call at +91 78922 39793. You can also send us a quick email at connect@cambrianbioworks.com
For immediate assistance, feel free to give us a direct call at +91 78922 39793. You can also send us a quick email at connect@cambrianbioworks.com